 To add the C4 term, see Tutorial 2.4 in the 12-6-4 nonbonded section. The parameters listed below contain an added r -4 term and reproduce the experimental hydration free energy (HFE), ion-oxygen distand (IOD) and coordination number (CN) values without significant compromise. 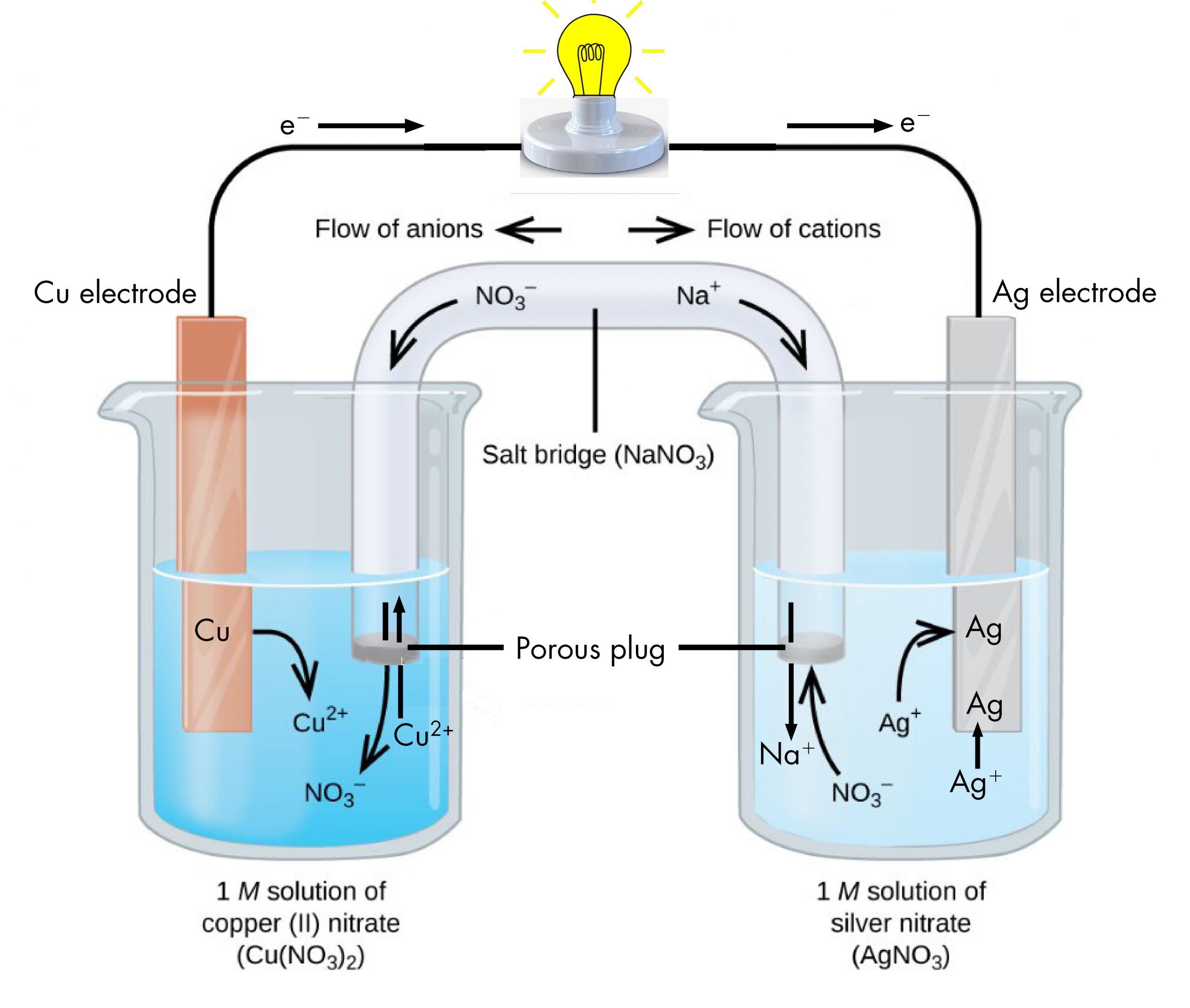
These ion parameters were parameterized against the experimental hydration free energies and/or ion-oxygen distances. Li +,Na +, K +,Rb +,Cs +,Tl +,Cu +,Ag +,NH 4 +,H 3O +, F -,Cl -,Br -,I -ĭivalent, trivalent and tetravalent also available - see manual Load the ions lib file and matching water-model frcmod file: This consistent set of parameters for alkali halide ions were developed by fitting solvationįree energies, radial distribution functions, ion-water interaction energies and crystal lattice energies and latticeĬonstants for non-polarizable spherical ions. Ion Parameter Set Availability and Implementation Joung and Cheatham 1 To use the 12-6-4 model see Tutorial 2.4 in the 12-6-4 nonbonded section. One needs to source the leaprc file for the water and then load the 12-6-4 frcmod file by itself, and then use ParmEd to add the C4 terms. To use the 12-6-4 parameters requires an extra step to insure the C4 term is implemented. However ion parameters are chosen, they should be consistent with each other (all 12-6 or all 12-6-4) and the water model.įor highly charged metal ions, the 12-6-4 LJ-type nonbonded model performs much better than the You should read each article and understand the pros and cons of each. Some ions are parameterized against hydration free energies (experimental or TI calculations), ion-oxygen distances and/or lattice energies. Ideally the ions are chosen to be compatible with the water model. ) will require a set of parameters (lib and frcmod file). Just like each type of biomolecule, each ion (Na +, K +, Mg 2+, Cl -, etc. If the anion is a simple ion it takes its name from the root name of the element, and -ide is added.For a full list and explanation of different ion parameters,Ĭhoosing parameters for ions can be tricky. However, since we only talk about one polyatomic type II cation this should not prove to be difficult. If the cation is a polyatomic you use the name that you have memorized. If the cation is a "simple" cation the cation is simply the name of the element. For example ferric is Fe 3+ and ferrous is Fe 2+. ic is for the higher charged cation, -ous is for the lower charged cation. Occasionally suffixes, -ic and -ous, are used however, we will not be using them. The cation is named first and the charge of the cation is expressed as a Roman numeral. If the cation is not from group 1 or 2, or is not Ag +,Cd 2+, Zn 2+, or Al 3+ then assume the cation is a type II cation. Naming compound with type II cations-type II cations are capable of having several charges. NaCl, MgCl 2, AlCl 3, NaNO 3, BaSO 4, (NH 4) 2SO 3ī. If the anion is a polyatomic anion you use the name that you have memorized. If the anion is a simple ion it takes its name from the root name of the element, and "-ide " is added. The Hg 2 2+ cation is a polyatomic ion and a type II cation.). If the cation is a polyatomic cation (a polyatomic ion is made from two or more covalently bonded elements.) you use the name that you have memorized (however, since we only talk about a couple of polyatomic cations this should not prove to be too difficult. 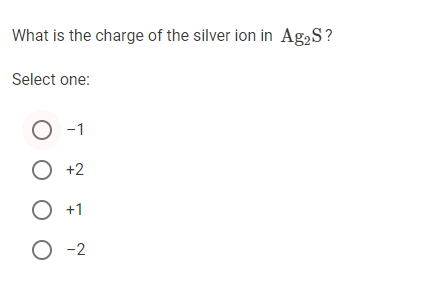
If the cation is a "simple" cation ("simple" ion is made from only one element.) the cations is named after the element. A few of the elements in groups 3 though 13 form only one cation therefore, a few of them are type I cations-examples include Ag +, Cd 2+, Zn 2+, and Al 3+. All the elements in the first two families form type I cations. Naming compounds with type I cations-type I cations are cations that have one possible charge. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
